
112 Drug Distribution and Control: Preparation and Handling–Guidelines
Purpose
The compounding of medications is a fundamental part of
pharmacy practice. All compounding personnel, mainly
pharmacists and pharmacy technicians, are responsible for
compounding and dispensing sterile products and prepara-
tions of correct ingredient identity, purity (freedom from
physical contaminants, such as precipitates,
1
and chemical
contaminants), strength (including stability
2
and compat-
ibility), and sterility and for dispensing them in appropriate
containers that are labeled accurately and appropriately for
the end user. In contemporary health care organizations, pa-
tients receive compounded sterile preparations (CSPs) that
are stored for extended periods before use. It has long been
recognized that extended storage of CSPs may allow for the
growth of a pathological bioburden of microorganisms
3
and
that patient morbidity and mortality can result from con-
taminated or incorrectly compounded sterile preparations.
4–9
When quality monitoring is inadequate, personnel respon-
sible for sterile compounding may not know that inaccurate
or contaminated products are dispensed.
10–13
These guidelines are intended to help compounding
personnel prepare CSPs of high quality and reduce the po-
tential for harm to patients and consequences for compound-
ing personnel. The recommendations in these guidelines are
based on published data, when available; on expert opinion
and procedures used in similar industries; and on applica-
ble regulations and standards. These guidelines are a revi-
sion of the 2000 ASHP Guidelines on Quality Assurance of
Pharmacy-Prepared Sterile Products,
14
with the goals of
providing more current recommendations and harmoniz-
ing the ASHP guidelines with United States Pharmacopeia
(USP) chapter 797, Pharmaceutical Compounding—Sterile
Preparations.
15
To help achieve that harmonization, these
guidelines employ the definitions and terminology of USP
chapter 797 rather than those of the previous guidelines.
Many health care settings also use CSPs prepared
by compounding pharmacies. Although these guidelines
may be useful in assessing the quality of CSPs prepared by
compounding pharmacies, more information on the topic
of outsourcing sterile compounding services is available in
the ASHP Guidelines on Outsourcing Sterile Compounding
Services.
16
Finally, while these guidelines are generally appli-
cable to all personnel who prepare CSPs and all facilities
in which CSPs are prepared, pharmacists and other health
care professionals responsible for the preparation, selection,
and use of CSPs are urged to use professional judgment in
interpreting and applying these guidelines to their specific
circumstances. Users of these guidelines are cautioned that
the information provided is current as of publication and are
urged to consult current editions of original sources (e.g.,
laws, regulations, and applicable standards, including USP
compendial standards) to ensure patient safety as well as le-
gal and regulatory compliance.
ASHP Guidelines on
Compounding Sterile Preparations
Legal and Regulatory Considerations
Significant legal and regulatory changes have taken place
since publication of the previous ASHP guidelines (Figure 1).
At the time of its publication, section 503A of the
U.S. Food and Drug Administration Modernization Act
(FDAMA) served to define the limits of legitimate com-
pounding.
18
When section 503A of FDAMA was ruled un-
constitutional in 2001, the delineation between compound-
ing and manufacturing reverted to earlier regulations based
on the Federal Food, Drug, and Cosmetics Act.
19
Under
those regulations, compounding is considered part of the
practice of pharmacy and in most states, is governed by state
law and regulation. Manufacturing is regulated by the fed-
eral government through the auspices of the Food and Drug
Administration (FDA). In most cases, extemporaneously
compounded preparations must be prepared pursuant to a
prescriber’s prescription for a specific patient. Some states
have specific regulations dealing with CSPs for office use.
Some pharmacies whose primary purpose is preparing CSPs
for hospitals and other facilities may be registered with the
FDA as manufacturers and must adhere to federal good
manufacturing practices. Some state boards of pharmacy
permit one pharmacy to compound for another pharmacy
under central fill regulations. Most pharmacies compound
only pursuant to a prescriber’s prescription and follow state
regulations regarding compounding.
On January 1, 2004, USP chapter 797, Pharmaceutical
Compounding—Sterile Preparations,
15
became official, re-
placing USP chapter 1206, Sterile Drug Products for Home
Use.
20
The change from a chapter numbered above 1000 to a
chapter below 1000 marked a change from an advisory stan-
dard to an enforceable one. USP chapter 797 has since been
revised.
15
Some state regulations require full compliance
with USP chapter 797, some have indirect references to the
chapter, some do not mention the chapter, and some have ad-
ditional regulations.
21
The National Association of Boards of
Pharmacy supports the incorporation of compounding regu-
lations into state pharmacy practice legislation by including
such wording in the association’s Model Rules and Model
State Pharmacy Act.
22
State boards of pharmacy should be
consulted to determine the current status of sterile com-
pounding regulations, as there are significant differences in
regulation among states.
Accreditation Considerations
The Centers for Medicare and Medicaid Services (CMS)
Hospital Conditions of Participation and Interpretive
Guidelines, the Joint Commission, the American Osteopathic
Association’s Healthcare Facilities Accreditation Program,
and DNV Healthcare’s National Integrated Accreditation
for Healthcare Organizations all include statements con-
cerning safe practices for storage and preparation of sterile
compounds.
23–26
Clinics, long-term care facilities, home care
organizations, rehabilitation facilities, and physician offices
(all of which come under the purview of USP chapter 797
15
)
Drug Distribution and Control: Preparation and Handling–Guidelines 113
may all be subject to specific additional governance of ster-
ile compounding practices, depending on the agencies regu-
lating or accrediting the facility. In addition, organizations
preparing hazardous drugs
27,28
should comply with National
Institute for Occupational Safety and Health (NIOSH) rec-
ommendations to ensure that compounding personnel are
operating in a safe environment.
29,30
Other Compounding-Related Guidelines
ASHP provides several guidelines for safe compounding
practices
28,31,32
and a discussion guide on USP chapter 797
17
and has recognized USP chapter 797 as a relevant practice
standard in the ASHP Guidelines: Minimum Standard for
Pharmacies in Hospitals.
33
Other professional organizations
also provide guidance on specific aspects of compounding.
Standards for prescribing, preparation, administration, and
monitoring of parenteral nutrition are available through the
American Society for Parenteral and Enteral Nutrition.
34,35
The Institute for Safe Medication Practices provides rec-
ommendations for preventing medication errors, including
those involving CSPs.
36,37
The Infusion Nurses Society of-
fers standards, professional development, and resources for
all aspects of infusion care.
38
The Controlled Environment
Testing Association (CETA) provides numerous CETA
Application Guides (CAGs) for the proper use, cleaning,
and certification of primary engineering controls (PECs)
and buffer areas (generally referred to as “cleanrooms”).
39–45
Guidelines for Hand Hygiene in Healthcare Settings,
46
Guidelines for Prevention of Intravascular Catheter-Related
Infections,
47
Guidelines for Environmental Infection Control
in Healthcare Facilities,
48
and Protect Patients Against
Preventable Harm from Improper Use of Single-dose/
Single-use Vials,
49
all from the Centers for Disease Control
and Prevention (CDC), serve as the backbone for most infec-
tion prevention practices in the United States. Safe infusion,
injection, and medication vial practices have been addressed
by CMS
50
and the Association for Professionals in Infection
Control and Epidemiology,
51
and the Association of peri-
Operative Registered Nurses has recommended practices for
medication safety in perioperative settings.
52
Physical Facilities and Equipment
Design and Functionality Requirements
Facility requirements are intended to establish a safe en-
vironment for compounding CSPs. The International
Organization for Standardization (ISO) air cleanliness clas-
sification of the compounding environment is a critical mea-
sure that is affected by facility design.
Primary Engineering Controls (PECs). A PEC is a device
or room that provides an ISO Class 5 environment for com-
pounding CSPs. PECs all rely on a special type of high-
effi
ciency particle air (HEPA) filter that is >99.99% efficient
in removing particles as small as 0.3 microns in size (the
most penetrating particle size [MPPS], which refers to the
largest-sized particle that may escape the filter, although
particles of all sizes may be captured). The unidirectional
(horizontal or vertical) HEPA-filtered air must provide
sufficient velocity to sweep particles away from the direct
compounding area and maintain unidirectional flow during
preparation of CSPs. (More information about HEPA filtra-
tion and first-air concepts can be found in the ASHP publica-
tions Compounding Sterile Preparations,
53
Basics of Aseptic
Compounding Technique,
54
Getting Started in Aseptic
Compounding,
55
and Compounding Sterile Preparations:
ASHP Video Guide to USP <797>.
56
)
PEC devices include laminar airflow workbenches
(LAFWs), biological safety cabinets (BSCs), compounding
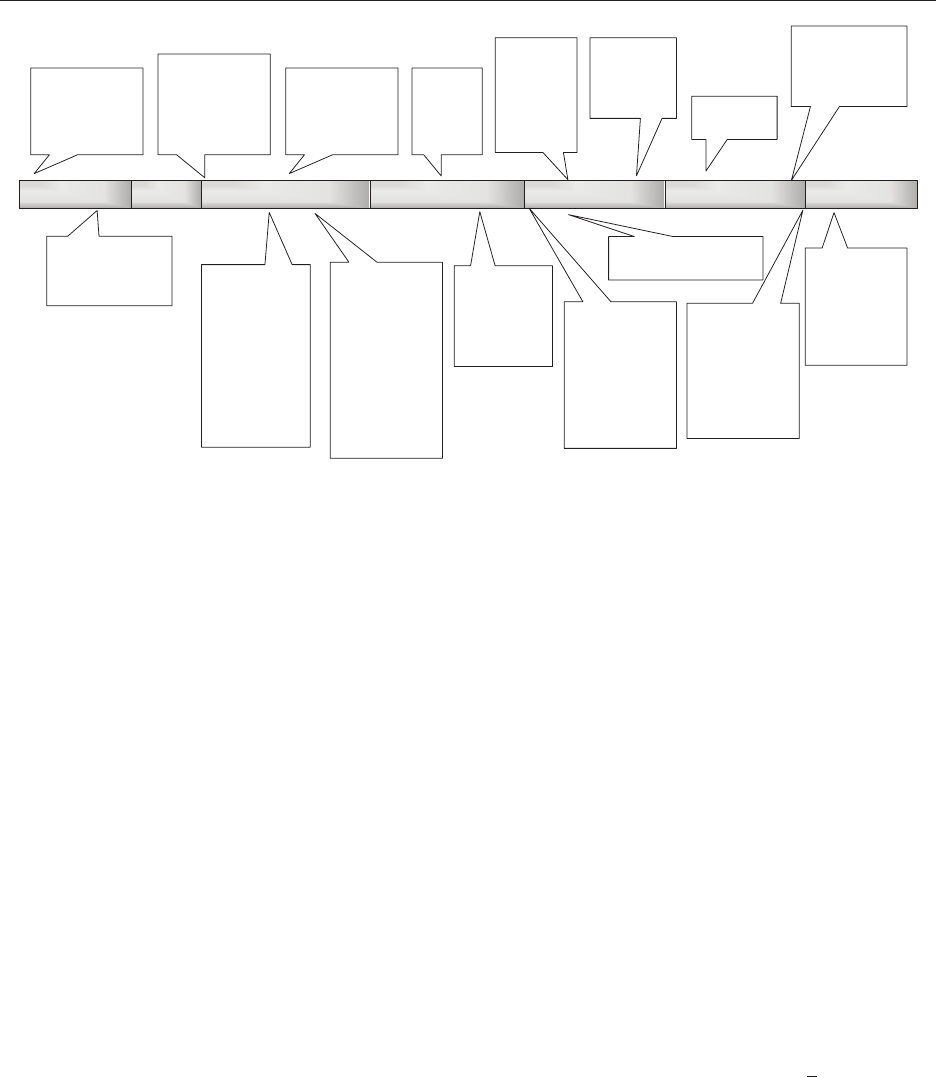
Figure 1. Evolution of Sterile Compounding Standards, 1970—2010. Adapted from The ASHP Discussion Guide on USP Chapter <797> for
Compounding Sterile Preparations.
17
NCCLVP, National Coordinating Committee on Large Volume Parenterals; CSPs, compounded sterile
preparations; USP, United State Pharmacopeia; TAB, technical assistance bulleting; FDAMA, Food and Drug Administration Modernization Act;
PCAB, Pharmacy Compounding Accreditation Board.
1970 20052000199519901980 2010
1971—Deadly
nationwide
nosocomial
infection
outbreak
1992—FDA
Compliance
Guide, USP
Dispensing
Practices for
Sterile Drug
Products
Intended for
Home Use
published
1991 and 1995—
ASHP national
compounding
surveys
1990—4 deaths,
2 cases of
blindness from
contaminated
CSPs
1975−80: NCCLVP
publishes
recommendations
1993—ASHP
TABs on Quality
Assurance for
Pharmacy-
Prepared Sterile
Products and on
Pharmacy-
Prepared
Ophthalmic
Products
published
1997—
FDAMA
signed
into law
USP Sterile
Compounding
Expert
Committee
formed
2000—ASHP
Guidelines on
Quality
Assurance for
Pharmacy-
Prepared Sterile
Products
published
2004—USP
Chapter 797
becomes
official
2002—
FDAMA
section 503A
ruled
unconsti-
tutional
2002—ASHP national
compounding survey
2006—PCAB
established
2008—USP
Chapter 797
revisions become
official
2012—
contaminated
CSPs injure
271 and cause
21 deaths in 16
states
2010—ASHP
Guidelines on
Outsourcing
Sterile
Compounding
Services
published

114 Drug Distribution and Control: Preparation and Handling–Guidelines
aseptic isolators (CAIs), and compounding aseptic contain-
ment isolators (CACIs) (Table 1). Properly designed, uni-
directional airflow CAIs function in a similar manner as
LAFWs, but the direct compounding area does not interact
with room air because it is within a closed system, with the
air sweeping particles away from the compounding site.
Smoke tests of PECs assist a facility in verifying unidirec-
tional airflow and lack of turbulence and reverse flows.
CAIs or CACIs located outside of an ISO Class 7 en-
vironment must be coupled with documentation from the
manufacturer that the device will meet or exceed USP chap-
ter 797 standards under these conditions and be dynamically
tested on site to USP 797 and CETA requirements. If the
CACI used for hazardous drug preparation is located outside
the buffer area (see Architecture, below), it must be located
in a segregated and dedicated area that maintains at least
0.01-inch water column negative pressure and maintains, at
a minimum, 12 air changes per hour (ACPH).
Architecture. The sterile compounding area includes a well-
lit buffer area and ante area (both are secondary engineering
controls) and an area for storage of sterile products and sup-
plies. A buffer area (or “cleanroom”) is defined as an area
where a PEC is located and where activities such as prepa-
ration, compounding, and staging of CSPs occur. This area
should provide adequate space for the PEC and may include
a limited amount of shelving and/or carts for staging of com-
pounding (not for storing stock). An ante area provides space
for hand washing, garbing, and product decontamination;
it also serves as a way to further segregate the buffer area
from other, less-clean areas of the facility. Water sources,
such as sinks or floor drains, are not permitted in the buffer
area and should not be immediately adjacent to segregated
compounding areas outside of a buffer area. A storage area
outside the buffer and ante areas should provide adequate
space for placement of sterile products and supplies.
The sterile compounding area (ante and buffer areas)
may be constructed of either hard- or soft-walled enclosures,
with the zones being delineated by open or closed architec-
ture. Closed architecture is formed by walls and doors be-
tween the buffer and ante areas and is required for high-risk
compounding (Table 2).
Open architecture has openings between the buffer and
ante areas and relies on a defined airflow velocity to divide
the two areas, which are marked by a line of demarcation;
this type of facility may only be utilized for low- and me
-
dium-risk compounding. Demarcation lines should be indi-
cated by colored tiles or other elements integrated into the
flooring pattern but may be as simple as marking on the floor
.
Facilities for preparation of radiopharmaceuticals have
some different requirements. Refer to USP chapter 797
15
and
other relevant standards for specifics.
Facilities without USP chapter 797-compliant ante ar-
eas and buffer areas may prepare low-risk, non-hazardous
CSPs in a PEC within a segregated compounding area. A
segregated compounding area is an unclassified space (i.e.,
an area with no specific ISO classification) and does not in-
clude ante or buffer areas. It is required to be separated from
activities that are not essential to the preparation of CSPs;
not be located adjacent to food preparation sites, ware-
houses, or construction sites; and not have unsealed win-
dows or doors that connect to the outdoors or high-traffic
areas.
15
This architecture type is most often seen in satellite
pharmacies, small hospitals, procedural areas, or clinics. The
beyond-use dating for sterile preparations compounded in a
segregated compounding area cannot exceed 12 hours (see
Expiration and Beyond-Use Dating).
Buffer Areas
Air Supply. A buffer area differs from an ordinary ventilated
room by having the following:
•
Increased air supply.
•
HEPA filtration (the filtered air should be introduced
at the ceiling, with returns mounted low on the walls;
ceiling-mounted returns should not be used) including
a terminal air filter (a filter at the end of the heating,
ventilation, and air conditioning [HVAC] ducting).
•
Room pressurization.
•
A perforated plate or swirl supply air diffuser (if an air
diffuser is necessary); high-induction supply air dif-
fusers should not be used in buffer areas.
Structural components must be coupled with HEPA
filtration and air exchanges in order to provide a complete
buffer area environment and proper ISO classifications.
Buffer areas must meet or exceed ISO Class 7 air cleanli-
ness standards. Ante areas must at least meet ISO Class 8
standards; ante areas opening into a negative pressure prepa-
ration area must meet ISO Class 7 standards. The number of
ACPH is based upon air/room pressure, velocity or air han-
dler capacity, HEPA flow restriction, duct size, the amount
of processing completed on a daily basis, and temperature.
ACPH must occur at a minimum of 30 times per hour in
buffer and ante areas, but may need to be increased in high-
traffic/high-volume areas in order to maintain the room’s
specified ISO classification (Table 2) under dynamic condi-
tions. Facilities may incorporate the contribution of up to 15
air changes per hour from a LAFW in the total air changes
per hours in a nonhazardous buffer area. By design, these
devices filter room air as it passes through the HEPA filter.
Airflow within the room should be as steady as pos-
sible, having as few interruptions as possible. Within the
PEC, it must be unidirectional,
39
with as few interruptions in
steady airflow as possible. PEC placement within the room
should be well designed, with PECs placed where they are
least affected by opened doors, HVAC systems, or personnel
traffic. For non-hazardous preparations, positive pressure
is required between rooms physically divided by walls or
doors (closed architecture style) and should be maintained
at a minimum of positive 0.02 inch water column. If a room
does not have physical barriers (i.e., has an open architecture
Table 1.
Primary Engineering Controls (PECs)
PEC Device
Used to Prepare
Non-Hazardous
CSPs
Used to Prepare
Hazardous CSPs
Conventional Laminar airflow
workbench
(LAFW)
Class II Biological
safety cabinet
(BSC)
Isolators Compounding
aseptic isolator
(CAI)
Compounding
aseptic
containment
isolator (CACI)

Drug Distribution and Control: Preparation and Handling–Guidelines 115
style) and relies on a line of demarcation, the displacement
airflow concept requiring air velocity of 40 feet per minute
(0.2 meter per second) from the buffer area across the entire
plane of line of demarcation into the ante area is required.
Open architecture is not permitted in areas used for high-risk
preparations.
When designing buffer areas, facilities must con-
sider workflow patterns, such as how personnel performing
double-checks will affect air quality. If supervisory person-
nel are not located in the buffer area, movement in and out
of the buffer area is likely to increase airflow interruption.
Communication devices should be used to minimize traffic
between areas, and cameras may be installed to supplement
supervision of staff or check compounding accuracy, if per-
mitted by state regulations.
Surfaces. Surfaces of any kind in the buffer area and ante
area must be smooth, impervious, and easy to clean, with
no cracks or crevices that could trap dust or contaminants.
All materials used in the facilities must be non-shedding.
Walls and ceilings must be made of either hard plastic or
epoxy-painted gypsum board. If ceiling tiles are used, they
must be coated with hard polymer and caulked both around
the perimeter and around each tile. Ceiling lights must be
smooth, mounted flush, and sealed. Floors should be made
of wide, heavy-duty sheet vinyl, rubber, or epoxy that is
coved around the corners and rolled up onto the walls. Paint
must be an epoxy, acrylic, or other non-porous sealant type.
Work surfaces should preferably be stainless steel,
but at a minimum are required to be non-porous and eas-
ily sanitized. Carts and shelves, ideally made of stainless
steel wire, nonporous plastic, or rustproof metal, should be
easy to move and clean, if necessary. Office equipment (e.g.,
computers and components [including washable keyboard
and mouse], telephones, printers) placed in the buffer area
must be easily cleanable and placed in such a manner that
they have no material impact on the ISO air cleanliness clas-
sification of the area.
Renovations
To meet requirements for sterile compounding, many facili-
ties choose to renovate existing space rather than construct
new facilities. Whether designing a new area or retrofitting
one, the specific types (e.g., hazardous or nonhazardous) and
risk levels of CSPs that will be prepared in the area should
guide the facility design and construction. A plan for how
operations will continue without interruption should be de-
vised prior to construction.
Power and Other Utility Interruptions
The facility’s emergency management plan should include
steps to meet patient-care needs during time of utility in-
terruptions, including the need for CSPs. In some cases,
immediate-use procedures may be safely implemented to
meet some needs. Methods to identify and safely meet in-
terim compounding needs or address patient-care needs with
Table 2.
Facilities Features Required for Specific Types of Compounding (Data from
USP
Chapter 797
15
Except as Noted)
Low-Risk with
≤12-hour BUD
(Non-Hazardous)
Low-Risk
(Non-Hazardous)
Medium-Risk
(Non-Hazardous)
High-Risk
(Non-Hazardous) Hazardous Drugs
Architectural Style
a
Segregated Open or closed Open or closed Closed Closed
Buffer zone ISO
classification
N/A ISO Class 7 or
better
ISO Class 7 or
better
ISO Class 7 or
better
ISO Class 7 or
better
Ante area ISO
classification
N/A ISO Class 8 (ISO
Class 7 if opens
into negative
pressure area)
or better
ISO Class 8 (ISO
Class 7 if opens
into negative
pressure area)
or better
ISO Class 8 (ISO
Class 7 if opens
into a negative
pressure area)
or better
ISO Class 7 or
better
Minimum air
exchanges for
buffer area
b
N/A 30 30 30 30
Minimum air
exchanges for
ante area
c
N/A 20 if ISO 8; 30 if
ISO 7
20 if ISO 8; 30 if
ISO 7
20 if ISO 8; 30 if
ISO 7
30
Pressure N/A Positive Positive Positive Negative
a
Architectural style (“open” and “closed”) is not defined in USP chapter 797, but the concept of physical separation of ante areas and buffer
rooms is described in the chapter. For the purposes of these guidelines, “closed architecture” indicates that the buffer and ante areas are
separated by a door (i.e., are physically separate rooms) and maintain a pressure differential of no less than 0.02-inch water column positive
pressure. “Open architecture” indicates that the buffer and ante areas are in one room, not separated by a door (i.e., not physically separated).
Displacement airflow is used to separate open architecture spaces, with at least 40 feet per minute of airflow across the entire plane of the
opening. A segregated compounding area contains a PEC within a restricted space.
b
If an ISO Class 5 recirculating device is in place, a minimum of 15 air changes per hour (ACPH) is sufficient if the ACPH is 30 between the
device and the area supply HEPA filters.
c
USP chapter 797 does not address the air changes in ISO Class 8 ante areas. The FDA Aseptic Processing Guide
57
recommends a minimum
of 20 ACPH to maintain ISO 8. However, this is a minimum value intended for industry. Since ante areas for CSPs include ungowned personnel and
other activities, a minimum of 30 ACPH is best practice for ISO Class 8 ante areas and required for ISO 7 ante areas.

116 Drug Distribution and Control: Preparation and Handling–Guidelines
noncompounded alternatives should be developed, put into
standard operating procedures (SOPs), inserviced to staff,
and tested as part of the organization’s emergency planning
process.
Pharmacy Compounding Devices
Pharmacy compounding devices are utilized to increase
efficiency while decreasing the potential for human error.
Devices that do not create their own ISO Class 5 environ-
ment must be located within an ISO Class 5 PEC and adhere
to applicable standards for accuracy and precision. All com-
pounding devices must be monitored and validated for ac-
curacy consistent with device manufacturer specifications.
Automated Compounding Devices (ACDs) are uti-
lized to accurately combine multiple drugs and solutions
into a single delivery container. These devices are most com-
monly used for parenteral nutrition preparation, but may be
used for cardioplegia solutions, continuous renal replace-
ment therapy, or other complex processes. ASHP Guidelines
on the Safe Use of Automated Compounding Devices for the
Preparation of Parenteral Nutrition Admixtures
32
should be
consulted for further details on utilizing ACDs. Accuracy
and precision testing for ACDs is required by USP chapter
797
15
and incorporate gravimetric, volumetric, and chemical
analyses. These analyses, as determined by facility protocol,
must be monitored and recorded on a daily basis, with evalu-
ation for outliers occurring at least weekly.
Repeater pumps are devices used to pump a preset vol-
ume of fluid in a consistent and reproducible manner. They
must be calibrated according to manufacturer specifications,
which may depend on the volume and frequency of use.
Robotic systems automate the compounding and label-
ing of parenteral doses in syringes and bags using an en-
closed chamber that must create an ISO Class 5 air cleanli-
ness environment or better.
The proper use of ACDs, repeater pumps, robotic sys-
tems, and other compounding equipment used in the prepa-
ration of CSPs remains the responsibility of the pharmacist.
Cleaning and Disinfecting
Cleaning with a germicidal detergent and water will remove
visible solids or soiling before disinfecting. Disinfecting
removes microbial contamination. It is critical that an ap-
propriate germicidal detergent and water be used to clean
all surfaces of the buffer and ante areas in addition to all of
the PECs. Great care must be exercised to avoid getting the
HEPA filters wet during cleaning. Cleaning with a germi-
cidal detergent will leave a residue that needs to be removed
from work surfaces (e.g., counter and PEC surfaces). This
residue is best removed by using sterile 70% isopropyl al-
cohol (IPA).
Appendix II of USP chapter 797
15
provides infor-
mation on types of products that can be used for cleaning
and disinfecting the ante and buffer areas, including floors,
walls, and ceilings. Choice of cleaning and disinfection
products should be approved by the organization’s appropri-
ate authority (e.g., the Infection Control Committee).
Policies and procedures must be developed to ensure
consistent practices, including dilution of cleaning products.
Table 3 describes the minimum frequency for cleaning sur-
faces used to compound low- and medium-risk CSPs in the
sterile compounding area.
Environmental Monitoring
Environmental monitoring and related documentation must
be completed on a routine basis to ensure adequate environ-
mental and personnel controls are in place to prevent con-
tamination of CSPs. Ensuring a safe compounding environ-
ment requires viable and nonviable airborne particle testing,
pressure differential or displacement airflow measurement,
temperature monitoring, and surface disinfection sampling
and assessment. Nonviable particles are particles that do not
contain a living organism, such as particles shed from pa-
per or dust. Viable particles are living organisms, such as
bacteria or fungal spores, that require nonviable particles to
travel. Monitoring of humidity,
39,44
sound,
39
and lighting
39
may also be considered by facilities to enhance the environ-
mental monitoring program.
Each element of the monitoring program must be
included in a sampling plan with sample locations, meth-
ods of collection, sampling frequency, and other specifics
depending on the type of monitoring being performed. The
environmental monitoring sampling frequency must occur
at a minimum as listed below, with possible additional times
based on the type of testing:
•
At the commissioning and certification of new facili-
ties and equipment.
•
Every six months during routine re-certification of
equipment and facilities.
•
After any facility or equipment maintenance, includ-
ing construction or remodeling of adjacent depart-
ments or work on shared air handlers.
•
At any point when problems are identified with prod-
ucts, preparations, or employee technique or if a CSP
is suspected to be the source of a patient infection.
Records of data collected through the monitoring pro-
gram must be maintained as part of the overall quality assur-
ance program of the facility. The data should be reviewed by
management personnel or their designees and by the facil-
ity’s Infection Control Committee to ensure that the findings
of the reports are addressed. Table 4 provides an overview of
environmental monitoring requirements.
Table 3.
Minimum Frequency for Cleaning of Specific Sites
(Reprinted with Permission from
USP
Chapter
797
15
)
Site Minimum Frequency
ISO Class 5 PEC Beginning of each shift
Before each batch
Every 30 minutes when
compounding
After spills
When surface contamination
is known or suspected
Counters and easily
cleanable work
surfaces
Daily
Floors Daily
Walls Monthly
Ceilings Monthly
Storage shelving Monthly

Drug Distribution and Control: Preparation and Handling–Guidelines 117
Temperature Monitoring. Any controlled temperature area
used for compounding sterile preparations or for storage of
sterile products or CSPs must be monitored at least once
daily and results documented in a log. The facilities should
maintain a comfortable room temperature (20 °C [68 ºF] or
cooler) for properly garbed compounding personnel. If fa-
cilities use continuous temperature recording devices, they
must be monitored and documented once daily to ensure
they are functioning properly. Controlled temperature ranges
are listed in Table 5.
Pressure Differential or Air Displacement. Since positive-
and/or negative-pressure rooms are required for sterile
compounding, the appropriate differential pressure or air
displacement velocities must be maintained. If closed archi-
tecture is used, a pressure differential between general, ante,
and buffer areas must be monitored. A facility with open
architecture design must monitor the differential airflow
across the opening between ante and buffer areas.
A pressure gauge or velocity meter must be in place to
monitor airflow between relevant areas. Pressure between
ISO Class 7 positive-pressure areas and the general area
must be at least 5 Pa (0.02-inch water column). Negative
pressure areas should have no less than 2.5 Pa (0.01-inch
water column) negative pressure to adjacent positive pres-
sure. A monitored pressure indicator must be installed to en-
sure proper pressurization. If differential airflow is used as a
measure, the velocity must be at least 0.2 meter per second
(40 feet per minute).
Results of pressure differential and/or velocity of air
displacement must be reviewed and documented each shift
(at least daily) or by a continuous device with alarms.
Nonviable Airborne Particle Testing Program.
Determination
of the ISO classification of an area or device is dependent
on nonviable particle testing (“certification”), which must
be completed by qualified personnel complying with the
Certification Guide for Sterile Compounding Facilities
(CAG-003-2006).
39
PECs such as LAFWs, BSCs, CAIs,
and CACIs must be certified every 6 months and whenever
the device is relocated or serviced. Both primary (LAFWs,
BSCs, CAIs, and CACIs) and secondary engineering con-
trols (buffer areas and ante areas) must be checked for total
particle counts every 6 months according to the manufac-
turer’s specifications or CETA recommendation and when a
device or room is relocated or altered. Thresholds for each
ISO class are presented in Table 6.
Viable Airborne Particle Testing Program. Classified space
(PECs and buffer and ante areas) must undergo routine vi-
able particle testing. The testing plan should include the re-
quired sample locations, method of collection, frequency,
the volume of air to be tested, and the time of day testing
will occur. Testing must occur every 6 months in all com-
pounding areas (PECs, buffer areas, ante areas, and areas
adjacent to segregated compounding areas) as part of the
overall compounding recertification process. The method
of testing must be impaction via an electronic air sampling
device, as settling plates alone are not considered an accept-
able method.
Sampling plans should be detailed and include all
high-traffic locations within the compounding area and any
sites prone to contamination. Turbulence caused by airflow
disruption, such as within an ISO Class 5 LAFW or door-
ways, should be included in the testing plan, along with ar-
eas where garbing, cleaning, labeling, and staging occur. In
segregated compounding areas, sampling should include lo-
cations within the ISO Class 5 PEC and other areas in close
proximity to the PEC.
Viable particle testing must be performed using a gen-
eral microbiological growth medium, such as sterile nutrient
Table 4.
Environmental Monitoring Requirements (Adapted from
USP
Chapter 797
15
)
Parameter Monitored By Frequency
Temperature Compounding personnel or facilities
management staff (if electronic monitoring is
centralized)
Documented daily (at a minimum)
Pressure differential or
velocity across line of
demarcation
Compounding personnel Documented each shift (preferably), daily (at a
minimum)
Qualified certifier At least every 6 months
Nonviable particles Qualified certifier At least every 6 months
Surface sampling Compounding or laboratory personnel Periodically, as defined by compounding and
infection control personnel, at least every
6 months or after significant changes in
procedures or cleaning practices
Electronic device sample of
viable particles
Compounding personnel or qualified certifier At least every 6 months
Table 5.
Controlled Temperatures (Data from
USP
General
Notices and Requirements
58
)
Storage Condition Centigrade Fahrenheit
Room temperature 20 to 25 °C 68 to 77 °F
Cold temperature
(refrigerated)
2 to 8 °C 36 to 46 °F
Freezer (frozen) −25 to −10 °C –13 to 14 °F

118 Drug Distribution and Control: Preparation and Handling–Guidelines
agar. In facilities that compound high-risk preparations, test-
ing must also be done with a medium that supports fungal
growth, such as malt extract. The growth medium should be
incubated (outside of the sterile preparation area) according
to the manufacturer’s recommendations.
Sample data must be reviewed as a means of evaluat-
ing control of the compounding environment. Results above
recommended action levels (see Table 7) should prompt
reevaluation of work practices, cleaning procedures, and
HEPA filtration. Any microbial growth that results from vi-
able environment sampling must be identified to the genus
level by microbiology personnel. If any highly pathogenic
organisms (e.g., gram-negative rods or yeasts) are identi-
fied, infection control specialists should immediately be
consulted to assist in formulating a response to the situation.
Surface Disinfection Sampling and Assessment. Touch
contamination originating from contaminated work surfaces
must be minimized and prevented if possible. Surface sam-
pling provides facilities with a snapshot of the effectiveness
of their disinfection procedures (including technique and
cleaning products) and must be part of the overall quality
assurance plan. Using a sterile nutrient agar contact plate
for flat surfaces or swabs for equipment and other non-flat
surfaces, sampling must be performed in all ISO classified
areas on a periodic basis, at a minimum, every 6 months or
when significant procedural or cleaning changes are imple-
mented. A specific plan detailing the location of each sam-
ple must be devised so that the same locations are repeated
with each testing session. Contact plates require pressing a
plate directly to the surface being tested, while swabbing re-
quires swabbing an area, submersing the swab in the correct
amount of diluent, and then swabbing onto or into a ster-
ile nutrient agar surface. Agar plates will leave a residue on
Table 6.
Particle Limits for Sterile Compounding Areas (Adapted from
USP
Chapter 797
15
)
Primary Engineering Controls
(LAFW, BSC, CAI, CACI)
Buffer Area and Ante-Area
Opening into a Negative
Pressure Room
Ante-Area Opening Only into
a Positive Pressure Room
ISO Class 5 7 8
Limit on number of ≥ 0.5
micron particles/m
3
of air
3,520 352,000 3,520,000
contact surfaces that must be cleaned with sterile water and
disinfected with sterile 70% IPA.
Results must be reported in colony-forming units
(CFUs) per plate. Reevaluation of work practices and clean-
ing procedures should occur if the CFU count exceeds the
suggested action levels (Table 8). Investigation into the
source of contamination should be undertaken, the sources
eliminated, and the area cleaned and re-sampled.
Environmental monitoring and quality assurance pro-
grams and documentation may be completed by a limited
number of personnel in any given facility, but the actions
of all compounding personnel may affect these two critical
elements of compliance. All compounding personnel should
be familiar with all facility policies and procedures specific
to CSPs, even if the procedures are not typically their re-
sponsibility.
Expiration and Beyond-Use Dating
A manufacturer’s expiration date is the date assigned pursu-
ant to manufacturer testing. The drug product is guaranteed
by the manufacturer to be safe and effective up to the listed
date when products are stored as described in the product
labeling.
A beyond-use date (BUD) is the date or time after
which administration of a CSP shall not be initiated. As
described in previous ASHP guidelines
14
and in USP chap-
ter 797,
15
the BUD is determined from the date or time the
preparation is compounded, its chemical stability, and the
sterility limits described later in these guidelines. Both the
stability of the components and the sterility limits described
above must be taken into consideration when determining
BUDs, and the BUD must be the shorter of the sterility dat-
ing or chemical stability dating. Information regarding sta-
bility dating procedures and defaults can be found in USP
chapter 795, Pharmaceutical Compounding—Non-Sterile
Preparations,
59
and other published literature sources.
60,61
Processes such as thin-layer chromatography (TLC)
and high-performance liquid chromatographic (HPLC) as-
says are the most reliable means of determining the stability
of a product and should be used in place of theoretical pre-
dictions of stability when published literature is not avail-
able. The use of commercial reference laboratories that of-
fer qualitative and quantitative testing may serve as a key
resource for end-product testing.
Risk Level Classification
In these guidelines, as in previous ASHP guidelines
14
and
USP chapter 797,
15
CSPs are stratified by potential risk of
Table 7.
Viable Environmental Monitoring Recommended
Action Levels for Microbial Contamination
(Adapted from
USP
Chapter 797
15
)
ISO
Classification
Recommended Action
Levels for Microbial
Contamination (CFUs/m
3
)
a
5 1
b
7 10
8 or above 100
a
CFUs/m
3
, colony-forming units per cubic meter.
b
Samples from ISO Class 5 environments should normally yield
no microbiological contaminants.

Drug Distribution and Control: Preparation and Handling–Guidelines 119
not required to be located within an ISO Class 7 buffer area.
The PEC must be separate from other operations, including
sinks and other water sources or drains, and away from un-
sealed windows or doors that connect to high traffic areas,
construction, warehouses, or food preparation areas. Distinct
labeling for conveying short BUDs should be considered.
Medium-Risk CSPs
This category encompasses preparations requiring more
complex compounding processes, including:
•
Multiple doses of sterile products combined or pooled
to prepare a product that will be administered either
to multiple patients (i.e., batching of syringes or large
volumes), or one patient on multiple occasions (e.g.,
preparation for use over several days).
49
•
More than three commercially available sterile prod-
ucts are used to produce the compound.
•
More complex compounding processes (e.g., total par-
enteral nutrition).
All requirements for low-risk compounding regarding
location and aseptic technique must be followed.
High-Risk CSPs
High-risk CSPs are those
•
Prepared from nonsterile ingredients, including manu-
factured products not intended for sterile routes of ad-
ministration.
•
Compounded using a nonsterile device prior to termi-
nal sterilization.
•
Containing nonsterile water that are stored for more
than 6 hours before sterilization.
•
Exposed to conditions worse than ISO Class 5 air
quality for longer than 1 hour, if they contain or are
compounded from sterile contents of commercially
manufactured products or CSPs without antimicrobial
preservatives.
•
Containing bulk ingredients whose chemical purity and
content strength are not verified by labeling and docu-
mentation from suppliers or by direct determination.
•
Prepared by compounding personnel who are improp-
erly garbed or gloved.
Presterilization procedures for high-risk level CSPs,
such as weighing and mixing, shall be completed in no
worse than an ISO Class 8 environment.
CSPs in this category must be terminally sterilized
before administration to patients. Terminal sterilization is
defined by the FDA as the application of a lethal process
microbial contamination into three primary categories: low-,
medium-, and high-risk CSPs, with an additional category
for CSPs intended for immediate use
15
and a sub-category
for low-risk CSPs intended for use within 12 hours.
15
The
potential risk is based on the danger of exposing multiple pa-
tients to microbial bioburden and based on microbial growth
factors influenced by product storage time, temperature and
product ability to support microbial growth, surface and
time exposure of critical sites, and microbial bioburden in
the environment. Compounding personnel must determine
the appropriate risk level and the appropriate BUD for use
based upon chemical stability and the potential for micro-
bial, physical, or chemical contamination during compound-
ing. In making a risk-level determination, compounding per-
sonnel must evaluate where the preparation is being made,
the number of components or the number of aseptic breaches
needed to compound the preparation, and the complexity of
the compounding process. When circumstances make risk-
level assignment unclear, guidelines for the more stringent
risk level should prevail. For examples and a comparison of
the risk levels, requirements, and BUDs to be used in risk-
level determination, see Table 9.
Low-Risk CSPs
This category encompasses simple admixtures involving
closed-system transfer, measuring, and mixing of three or
fewer commercially manufactured sterile products (includ-
ing the infusion solution). Low-risk compounding condi-
tions must include all of the following:
•
CSPs are compounded using aseptic technique within
an ISO Class 5 PEC (e.g., LAFW, BSC, CAI, or CACI)
that is located within an ISO Class 7 buffer area with
an ISO Class 8 ante area.
•
Each container, including the final container, may not
be entered more than twice to prepare the CSP.
•
Compounding is limited to aseptic manipulations of
disinfected containers using sterile needles and sy-
ringes.
Low-Risk CSPs for Use Within 12 Hours. Under limited
circumstances, sterile compounding may occur in a seg-
regated compounding area (such as a satellite pharmacy
or dedicated sterile compounding space) in which the ISO
Class 5 PEC is not located within an ISO Class 7 buffer area.
A segregated compounding area is a designated space, ei-
ther a demarcated area or room, in which compounding is
restricted to preparing low-risk, nonhazardous CSPs with a
beyond-use time of no more than 12 hours from the time of
preparation. All other requirements for low-risk CSPs must
be followed, with the exception that the ISO Class 5 PEC is
Table 8.
Recommended Action Levels for Personnel Testing (Adapted from
USP
Chapter 797
15
)
PEC Buffer Area Ante Area
Viable airborne particle testing action levels for
contamination (CFUs per cubic meter [1000
L] of air per plate)
>1 >10 >100
Surface sample contamination (CFUs per plate) >3 >5 >100
Glove fingertip sampling >3 N/A N/A
a
CFUs = colony-forming units.

120 Drug Distribution and Control: Preparation and Handling–Guidelines
filtrated units. All filters used to sterilize CSPs must undergo
filter integrity (bubble-point) testing.
Immediate-Use CSPs
The immediate-use category should be reserved for emergent
use or situations in which adhering to low-risk compounding
procedures would add additional risk due to delays in patient
care. Examples of such situations may include cardiopulmo-
(e.g., steam under pressure or autoclaving) to sealed contain-
ers for the purpose of achieving a sterility assurance level
of less than 10
-6
or a probability of 1 nonsterile unit per 1
million sterilized units.
57
For CSPs that are heat-labile and
cannot be processed as above, sterilization using an alterna-
tive method, such as a sterilizing grade 0.22 micron filter,
must be done. Filtration only achieves a sterility assurance
level of 10
-3
, which is only 1 nonsterile unit per one thousand
Table 9.
CSP Risk Levels and Beyond-Use Dates (BUDs) (Adapted from
USP
Chapter 797
15
)
a
Risk
Category
Compounding
Location
Garbing
Required
Aseptic
Technique
Required Examples
BUDs of CSP Stored at
Room
Temperature Refrigerated
Frozen
(≤ 10 °C)
Low-risk ISO Class 5
PEC, ISO
Class 7
buffer area,
ISO Class 8
b
ante area
Yes Yes Reconstitution of a
single-dose vial, single
preparation of a small
volume parenteral,
single large volume
IV replacement fluids
with no more than 3
components
48 hours 14 days 45 days
Low-risk
with
< 12-hour
BUD
ISO Class
5 PEC
segregated
from other
operations
Yes Yes Same as low-
risk examples,
non-hazardous
preparations only
12 hours 12 hours N/A
Medium-risk ISO Class 5
PEC, ISO
Class 7
buffer area,
ISO Class 8
b
ante area
Yes Yes Batched syringes,
total parenteral
nutrition, ophthalmic
preparations made
from sterile products,
pooled admixtures,
batch-compounded
preparations without
bateriostatic additives,
preparations made
using automated
compounders or other
automated devices,
elastomeric pumps
30 hours 9 days 45 days
High-risk ISO Class 5
PEC, ISO
Class 7
buffer area,
ISO Class 7
ante area
Yes Yes CSPs prepared from
bulk, nonsterile
components or in
final containers
which are not sterile;
preparations that must
be terminally sterilized
before administration
24 hours 3 days 45 days
Immediate-
use
Medication
preparation
areas should
be clean,
uncluttered,
and
functionally
separate
c
No Yes Emergent use
preparations such as
epidurals prepared
by anesthesia for
immediate injection or
infusion, diagnostics,
any non-hazardous
preparations that
might cause harm
due to delays in
administration
1 hour N/A N/A
a
ISO = International Organization for Standardization, PEC = primary engineering control, IV = intravenous.
b
Ante area must be ISO 7 if it opens into a negative pressure buffer area.
c
Source: The Joint Commission. MM.05.01.07, EP2.
24

Drug Distribution and Control: Preparation and Handling–Guidelines 121
nary resuscitation, diagnostic procedures, or short-stability
medications that must be prepared immediately before ad-
ministration outside health care facilities (e.g., in home infu-
sion or emergency care at the accident site or in an ambu-
lance). Immediate-use CSPs do not need to be compounded
in an ISO Class 5 environment and garbing and gowning are
not required, as long as all of the following criteria are met:
•
Hand hygiene per CDC recommendations.
46
•
Aseptic technique is followed.
•
No hazardous drugs are used.
•
Only simple transfer of no more than three sterile,
non-hazardous drugs in the manufacturer’s original
containers are involved in the compounding, and no
more than two entries into any one container occur.
•
No more than 1 hour elapses from the time compound-
ing commences to the time administration to the pa-
tient begins (although best practice dictates that there
are no intervening steps between compounding and
administration).
•
No batching or storage of CSPs occurs.
•
The preparation is labeled with patient identification,
names and amounts of all ingredients, name or initials
of preparer, and exact 1-hour BUD and time.
If CSPs prepared for immediate use are not admin-
istered within 1 hour, they must be properly discarded. All
medications must be labeled to meet regulatory and accredi-
tation standards and in accordance with facility policy.
Point-of-Care Activation Systems
Point-of-care (POC) activation systems (i.e., vial/bag sys-
tems) create a physical barrier between components (fluid
and medication) that can be activated to allow the compo-
nents to mix. These devices are designed to create a closed
system by which the end user activates the components just
prior to the administration of the medication. BUDs for
these products are based on the individual manufacturer’s
recommendations for labeling and dating. Table 10 provides
a summary of manufacturer-recommended BUDs for POC
systems at time of publication. To decrease potential for con-
tamination and errors, POC systems that will be attached and
stored for longer than 1 hour prior to activation should be
assembled (but not activated) by pharmacy staff within an
ISO Class 5 environment. Activation of the devices should
be completed at the point of care just prior to administration.
Ampuls, Single-Dose, and
Multiple-Dose Containers
Ampuls may not be reused or saved at any time during the
compounding process. To minimize particulate contamina-
tion, 5 micron filter straws or filter needles must be used
when withdrawing contents of ampuls. Refer to the drug
labeling for manufacturer’s recommendations concerning
filtration.
The environmental conditions in which drug vials are
entered determine the BUD for the CSP. Single-dose vials
are intended to be used to prepare single doses; however, in
times of critical need, contents from unopened single-dose/
single-use vials may be repackaged for multiple patients.
49
This repackaging should only be performed by qualified
health care personnel in accordance with the procedures de-
scribed in these guidelines and in USP chapter 797.
15
Pharmacy bulk packages (PBPs), a type of vial con-
taining many single doses,
65
must be considered a single-
dose vial for purposes of determining BUDs. Manufacturer’s
information for each PBP contains recommended BUDs,
which are usually between 4 and 8 hours.
Multiple-dose vials may be reused or saved up to the
manufacturer’s recommended BUD, if they are not opened
in a direct patient-care area and if facility policy does not
require a shorter period.
66
If there is no manufacturer recom-
mendation, multiple-dose vials may be reused or saved up
to a maximum of 28 days or for a shorter period dictated by
facility policy. Table 11 illustrates the dating for these prod-
ucts based on environmental conditions.
The person who first punctures a multiple-dose con-
tainer intended for re-use must note the BUD and other in-
formation required by facility policy (e.g., his or her initials)
on the vial or attached label. A label indicating “use by”
clarifies that the date is the BUD rather than the opening
date. If a vial lacks a BUD, it should not be used and should
be properly discarded.
Table 10.
Beyond-Use Date (BUD) at Room Temperature for Point-of-Care Activated Devices Assembled in ISO Class 5
Environment
a
Device Company BUD
b
Applicable Products
ADD-Vantage
62
Hospira 30 days from date diluent
removed from overwrap
Mini-Bag Plus
63
Baxter 15 days from date diluent
removed from overwrap
50 and 100 mL bags
Mini-Bag Plus
63
Baxter 30 days from date diluent
removed from overwrap
100 mL containers docked with the following drugs: cefazolin
1 g, cefuroxime (Zinacef) 750 mg, ceftriaxone (Rocephin)
1 g, aztreonam (Azactam) 1 g, piperacillin and tazobactam
(Zosyn) 3.375 g
addEASE
64
B. Braun 70 days When connected to 50 mL and 100 mL bags
56 days When connected to Excel 250 mL bags
a
Information is current as of January 2011. The manufacturer’s package insert should always be checked for the most current recommendation for
dating.
b
BUD for assembled but not activated system.

122 Drug Distribution and Control: Preparation and Handling–Guidelines
Batch Compounding and Sterility Testing
Use of CSPs stored for extended periods of time is guided by
the chemical stability of components and the sterility limits
of the CSP defined above. If medium-risk batches are pre-
pared and assigned a BUD within those limits, no sterility
testing is required. However, if those limits are exceeded,
each batch must be tested for sterility according to the re-
quirements of USP chapter 71.
67
Facilities that wish to store CSPs for periods longer
than those described above must complete sterility testing
for each batch to determine the extended BUD. Each batch
of any risk-level CSP intended for storage outside the limits
described above must be tested for sterility, according to the
requirements of USP chapter 71, Sterility Tests.
67
The results
must be evaluated along with stability data to establish the
extended BUD. The policies and procedures of the indi-
vidual facility must outline the processes used to determine
extended BUDs.
Batches of high-risk CSPs prepared as multiple-
dose vials intended for administration to multiple patients,
batches of high-risk CSPs exposed for more than 12 hours
to temperatures of 2 to 8 °C (36 to 46 ºF) or for more than 6
hours to temperatures above 8 °C (46 ºF) before sterilization,
or batches of more than 25 identical, single-dose, high-risk
CSPs must undergo sterilization and microbial and bacterial
endotoxin (pyrogen) testing prior to dispensing or adminis-
tration. Sterility testing, as outlined in USP chapter 71, must
be completed prior to dispensing or administration.
67
USP
Membrane Filtration, USP Direct Inoculation of the Culture
Medium, or another testing method that produces verifica-
tion results statistically comparable with those methods may
be utilized.
67
If sterility testing results are not received prior to dis-
pensing, procedures must be in place for daily observation
of the sterility test specimens, immediate recall of dispensed
CSPs, and notification of patients and their physicians if mi-
crobial or fungal growth is observed. An investigation into
the root cause of contamination must occur if sterility testing
is positive.
All high-risk CSPs prepared in batches of more than
25 units, with the exception of inhalation or ophthalmic
preparations, must be tested to ensure that they do not con-
tain excessive bacterial endotoxins, as described in USP
chapter 85, Bacterial Endotoxins Test,
68
and USP chapter
151, Pyrogen Test.
69
Endotoxin limits (reported in USP en-
dotoxin units/hour/kg or units/hour/m
2
), if established, are
included in the official monograph for the product or may
be found in other formula sources. If specific endotoxin lim-
its are not available, default guidance can be found in USP
chapter 85.
68
For high-risk preparations, batches of 25 or fewer
CSPs do not require sterility testing.
15
However, facilities
should consider sterility testing of such CSPs as part of their
quality assurance plans to ensure that proper procedures are
being followed.
Outsourced CSPs
Outsourcing the preparation of CSPs to pharmacies that
specialize in sterile compounding provides an option for
facilities that cannot or do not wish to prepare all or some
types of CSPs (e.g., radiopharmaceuticals, high-risk CSPs,
parenteral nutrition) in their own facility. Facilities consid-
ering outsourcing compounding should consult the ASHP
Guidelines on Outsourcing Sterile Compounding Services.
16
The decision to use CSPs prepared by outside compounding
pharmacies should be reviewed and approved by hospital
leadership,
23,70
and such use should only occur in accordance
with written policies and procedures.
Administration of CSPs
USP chapter 797 does not include any specifications for ad-
ministration or timing during this crucial period of the drug
delivery cycle. CDC provides the most comprehensive guid-
ance regarding administration of intravenous medications,
including administration times, frequency of infusion set
changes, use of filters, and prevention of catheter-related
infections.
38,47
Personnel
Personnel Responsibilities
The term compounding personnel refers to any individual
involved in compounding sterile preparations, regardless
of profession. Compounding personnel are responsible for
ensuring that CSPs are accurately identified, measured, di-
luted, and mixed and are correctly purified, sterilized, pack-
aged, sealed, labeled, stored, dispensed, distributed, and
disposed of if not used. Emphasis should be on the need to
Table 11.
Beyond-Use Dates for Ampuls, Single-Dose, and Multiple-Dose Containers (Adapted from
USP
Chapter 797
15
)
Container
Opened and Maintained within an ISO
Class 5 Environment
Opened Outside an ISO Class 5 Environment or
Taken from ISO Class 5 Conditions to Less Clean Air
Ampuls One time use; cannot be stored One time use; cannot be stored
Single-dose vials One time use, cannot be stored; contents
of unopened vial may be repackaged in
times of critical need
49
One time use; cannot be stored
Pharmacy bulk packages 6 hours
a
Not intended for use outside ISO 5 environment
Multiple-dose vials 28 days
a
28 days
a
a
Unless otherwise specified by manufacturer.

Drug Distribution and Control: Preparation and Handling–Guidelines 123
maintain quality standards for the control of processes, com-
ponents, and environments and for the skill and knowledge
of personnel who prepare CSPs.
Accurate identification and inspection of quality and
purity of non-sterile chemicals or non-sterile ingredients
are necessary for the integrity of the finished preparations.
Upon arrival from the manufacturer and subsequently after
opening, bulk packages should be inspected for breaks in
the package or closure integrity and for proper appearance,
color, odor, and texture.
If nonsterile ingredients are not official USP or
National Formulary products, compounding personnel must
require a Certificate of Analysis from the manufacturer to
accompany the products.
59
Once a product is received from
the manufacturer, the date of receipt must be clearly marked
on each package. If a manufacturer’s expiration date is not
provided, chemicals should be given a three-year BUD from
the time of opening unless inspection or testing deems the
product within drug monograph specification (if available)
to be used for a longer time.
59
Compounding personnel must have an understanding
of how combining different agents in a preparation may af-
fect bioavailability, compatibility (visual and chemical), pH,
and concentration effects. Factors that influence stability
(e.g., temperature, pH, sorption, photolysis, and chemical
degradation) must be carefully evaluated and supported by
references or appropriate testing.
Compounding personnel must understand and dem-
onstrate competency in aseptic technique and for the prod-
ucts and systems used in CSP preparation, such as needles,
syringes, administration sets, fluid containers, and com-
pounding devices. Aseptic principles and techniques are
explained in depth in Compounding Sterile Preparations
53
and demonstrated in Basics of Aseptic Compounding
Technique,
54
Getting Started in Aseptic Compounding,
55
and
Compounding Sterile Preparations: ASHP’s Video Guide to
Chapter <797>.
56
Personnel must understand the types of
PECs, HEPA filtration, and airflow concepts that are critical
to sterile compounding.
Policies should be developed in conjunction with em-
ployee health or infection control personnel to set thresh-
olds for health status fitness for compounding personnel.
Compounding personnel with weeping sores, rashes, con-
junctivitis, or respiratory infections must not participate in
compounding processes until these conditions resolve.
Hygiene and Garbing. Proper preparation for sterile, non-
hazardous drug compounding must include effective hand
hygiene and garbing procedures. To minimize the number of
particles introduced into the sterile compounding area and
to minimize the risk of bacteria, all outer jackets and sweat-
ers, visible jewelry, and cosmetics must be removed prior to
initiating the handwashing and garbing processes. Personal
electronic devices (e.g., cell phones, MP3 players) and any
associated attachments must be removed prior to hand hy-
giene and garbing and should not be used within the sterile
compounding area.
Hand hygiene must be performed prior to and after
gowning and includes:
•
Washing hands, under the fingernails, wrists, and up
to the elbow for 30 seconds with a facility-approved
agent.
•
Drying hands and arms with nonshedding disposable
towels or an electronic hand dryer.
•
Sanitizing hands with application of a waterless,
alcohol-based hand rub (ABHR) with persistent activ-
ity prior to donning sterile gloves.
Garbing occurs in the ante area and should be sequenced as
follows (from “dirtiest” to “cleanest”):
•
Don shoe covers, hair and beard covers, and a mask.
•
Perform hand hygiene.
•
Don gown, fastened securely at the neck and wrists.
•
Sanitize hands using an ABHR and allow hands to dry.
•
Enter the buffer area (if facility layout dictates, this
step may occur after the following two steps).
•
Don sterile powder-free gloves.
•
Sanitize the gloves with application of 70% sterile IPA
and allow gloves to dry.
Studies support the use of sterile rather than nonsterile
gloves in the reduction of initial bioburden.
71
Furthermore,
nonsterile gloves run the risk of cross-contamination from
hands touching multiple gloves as they are removed from a
stock box or container. Gloves must be inspected by person-
nel on a routine basis during the compounding process to
check for tears or holes. The gloves should be disinfected
with sterile 70% IPA throughout the compounding process
and each time contaminated items are touched.
When high-risk compounding operations prior to ter-
minal sterilization occur, personnel must glove and garb as
stated above.
When exiting the compounding area during a work shift,
gowns that are not soiled may be removed and retained in the
ante area and re-worn during the same work shift. All other
garb, including gloves, must be removed and replaced, and
proper hand hygiene must be completed before re-entering
the compounding area. When CAIs are utilized, compounding
personnel must glove and garb as above, unless the manufac
-
turer of the isolator provides written documentation based on
environmental testing
that any or all of the components of
personnel hygiene and garbing are not required based on the
PECs of the facility where the device is located.
Proper garb should always be used with CACIs, be-
cause personnel will be handling hazardous materials. Vials
may be contaminated, even upon delivery,
72–74
and the garb
is needed to protect compounding personnel from unex-
pected drug residue and from inadvertent spills.
Compounding Areas. Compounding personnel must under-
stand the purposes of and relationships between ante, buffer,
segregated, and storage areas. A systematic process of enter-
ing and exiting the various areas is necessary to minimize
contamination. Food, drinks, and gum are prohibited in all of
these areas. Since shedding from paper and labels provides a
source of nonviable particles, only paper products essential
to the compounding process should be allowed in the buffer
area. Corrugated cardboard packaging must be eliminated
from buffer areas and should be eliminated from ante areas,
with all products and components such as needles, syringes,
and tubing removed from their outer cardboard packaging
and decontaminated by wiping the individual packages (if
not in an overwrap) with a suitable disinfectant (e.g.,70%
IPA) prior to entering the buffer area.

124 Drug Distribution and Control: Preparation and Handling–Guidelines
When used for sterile compounding, items in plastic
or foil overwrap should remain in the overwrap until intro-
duced into the ISO Class 5 PEC, at which point they should
be opened immediately before placing in the PEC and the
overwrap immediately discarded.
75
Items stored in the buf-
fer area but not in an overwrap must be decontaminated
again prior to entering the PEC, as items may be stored in a
buffer area for an extended period of time and may become
contaminated by dust or other particles.
Packaging and Labeling
Packaging and subsequent labeling are critical to patient
safety. Packaging must be appropriate to preserve both ste-
rility and stability until the BUD. Proper labeling requires an
understanding of compounding risk levels and how to deter-
mine BUDs based on both stability and sterility.
Labels for single compounded preparations must, at a
minimum, include the following:
•
Names of active ingredients
•
Amounts or concentrations of active ingredients
•
BUD and time
•
Storage requirements
•
Identification of responsible compounding personnel
Labels for batch-prepared CSPs must also include:
•
Control or lot number
•
Appropriate auxiliary labeling (including precautions)
•
Device-specific instructions (when appropriate)
Federal and state regulations and accreditation re-
quirements may necessitate additional label information be-
fore the CSP is dispensed to a specific patient.
Verification of compounding accuracy and sterility
incorporates physical inspection, ensuring compounding ac-
curacy processes are in place, and (when applicable) steril-
ity and endotoxin testing. Finished preparation evaluation is
the responsibility of compounding personnel and should be
performed during the compounding process and when the
preparation leaves the storage area. Visual inspection should
assess particulate matter, coring, cloudiness, leaks, and con-
tainer and closure integrity.
Compounding accuracy checks must comply with fed-
eral and state dispensing regulations and include accuracy
of the product or preparation and the labeling. Prescription
orders, compounding procedures, records, and materi-
als used to prepare the compounds should be evaluated. A
process should be implemented to confirm that the com-
pounding process and end-preparation testing are properly
done. Checking procedures should follow facility policy
and procedures and may be accomplished via cameras or
other devices, by video recordings, or by keeping the used
additive containers and syringes with the final product until
checked. The check ideally should be performed by some-
one other than the compounder to decrease confirmation
bias. Accuracy can be further verified by weighing when ap-
plicable and practical. When using an ACD, specific gravity
values must be independently confirmed after being entered
to ensure proper volumes are delivered during the com-
pounding process.
Storage of CSPs
Temperatures of areas used for storage on patient-care and
procedural units, including room temperature and in refrig-
erators, freezers, and warmers, must be monitored and re-
corded daily. On at least a monthly basis, compounding per-
sonnel or designated pharmacy personnel should evaluate
storage areas for appropriate secure conditions, separation
of drugs and food, and proper use and disposal of single- and
multiple-dose vials.
Control and Oversight of IV Solutions
Some facilities delegate storage and distribution of paren-
teral solutions to materials management. Since the products
are prescription drugs, the pharmacy must maintain over-
sight, including selection of appropriate products, package
sizes, and forms; safe and secure storage; and temperature
control. IV solutions that contain medications (e.g., potas-
sium chloride, heparin, dopamine, dextran, mannitol) or
high-risk agents (e.g., sterile water, sodium chloride greater
than 0.9%, and parenteral nutrition components) should be
stored in and distributed by the pharmacy.
Transporting CSPs
All personnel involved in the handling, transport, or stor-
age of CSPs, whether they are compounding personnel or
not, must be properly trained to complete these tasks, and
the performance of all personnel, including contractors,
must be monitored for compliance with facility policies.
Transportation methods for CSPs should be evaluated, as
some forms of transportation, such as pneumatic tube sys-
tems, may adversely affect stability or integrity. Pneumatic
tube delivery may require additional padding around con-
tainers to ensure that heat and light exposure and impact are
minimized. Some preparations may degrade if shaken, and
therefore personnel, including pharmacy and nursing per-
sonnel, should be aware of which preparations may not be
delivered via a pneumatic tube device.
Hazardous drug transport must incorporate measures
to maintain CSP integrity while minimizing the risk of drug
residue exposure to patients, personnel, and the environ-
ment. These preparations should always be delivered in a
bag to prevent leakage or accidental exposure during trans-
port, and they should not be delivered using a pneumatic
tube device due to the risk of contamination to the environ-
ment if breakage occurs. Cleaning protocols for pneumatic
tube systems are inadequate for hazardous drug contamina-
tion throughout the system.
Transport may occur outside of the compounding facil-
ity to other facilities or directly to patients. In these situations,
compounding personnel must ensure physical integrity
, steril-
ity, and stability are maintained during transit. Proper packag-
ing must be chosen to prevent contamination, leaks, damage,
and temperature variations and to protect the end recipients
and transporting personnel from harm. Handling and expo
-
sure instructions should be legibly displayed on the outside of
shipping containers. BUDs, storage instructions, and disposal
instructions for out-of-date preparations must be available to
recipients, and recipients must be able to properly store CSPs
(e.g., in a refrigerator or freezer
, if necessary).

Drug Distribution and Control: Preparation and Handling–Guidelines 125
Redispensing CSPs
If facility policy allows redispensing of CSPs, the process
must only be done by compounding personnel to ensure
continued sterility, purity, and stability. Facilities must de-
termine how to track original preparation and thaw dates (if
applicable) and be able to detect product tampering. There
must be policies and procedures in place to provide assur-
ance of proper storage conditions for each product or prepa-
ration (e.g., refrigeration, protection from light, package in-
tegrity) before redispensing. CSPs must not be redispensed
if package integrity has been compromised, including tem-
perature variations.
Personnel Responsibilities for Handling,
Preparation, and Disposal of Cytotoxic
and Other Hazardous Agents
The Occupational Safety and Health Administration (OSHA)
requires that employers and employees be made aware of the
hazards of all chemicals used in the workplace, including
drugs.
76
Compounding personnel of reproductive capabil-
ity shall confirm in writing that they understand the risks of
handling hazardous drugs.
15
Personnel at high risk of expo-
sure to hazardous drugs should be enrolled in a medical sur-
veillance program. In many larger facilities, the employee’s
health department will determine who should be enrolled.
Specific guidance about surveillance for health care workers
exposed to hazardous drugs is available from NIOSH,
77
as
is a list of drugs NIOSH considers hazardous.
27
The risks of
occupational exposure to hazardous drugs and their poten-
tial effects on compounding personnel should be conveyed
to employees during employee orientation and in an ongo-
ing manner through continuing education and monitoring at
least annually. Training and competency programs should be
provided in addition to competencies for compounding of
non-hazardous sterile drugs, with details of differentiating
the garbing, storage, preparation, and disposal procedures
for hazardous drugs. USP chapter 797 requires that training,
at a minimum, include:
•
Safe aseptic manipulation practices.
•
Negative pressure techniques when compounding.
•
Proper utilization of a BSC or CACI.
•
Correct use of closed-system transfer devices
(CSTDs), if used.
•
Containment, cleanup, and disposal procedures for
breakages and spills.
•
Treatment of personnel contact and inhalation expo-
sure.
15
OSHA requires more general training on chemical
label elements and safety data sheet (SDS) format.
76
When
training or evaluating competency, facilities may choose
products to objectively evaluate hazardous drug compound-
ing technique. These products utilize dyes or fluorescence to
determine personnel technique and assess for spills or haz-
ardous drug exposures.
Definitions of hazardous drugs and proper handling
of hazardous drugs, including receiving, distribution, stock-
ing, inventorying, preparation, transport, and disposal, are
all concepts discussed in detail in the ASHP Guidelines on
Handling Hazardous Drugs.
28
Personnel Responsibilities
for Specialty Preparations
Specialty preparations (e.g., allergen extracts, radiopharma-
ceuticals, and evolving technology and therapeutics such as
biologics and nanotechnology) provide specific treatments
for patients and require specialized procedures to be fol-
lowed. Although compounding of intradermal or subcuta-
neous injections of allergen extracts would ideally occur
under conditions for similar risk-level CSPs, they may not
necessarily be subject to the same personnel, environmen-
tal, and storage requirements as for other CSPs of a similar
risk level, as long as the following criteria outlined by USP
chapter 797,
15
in conjunction with the American Academy
of Otolaryngic Allergy and the Joint Council of Allergy,
Asthma and Immunology,
78
are met:
•
Compounding process involves simple transfer using
sterile components and allergen products.
•
All allergen extracts contain effective preservatives to
prevent microbial growth.
•
All hand hygiene, garbing, and gloving procedures for
low-risk compounding, with the exception of donning
shoe covers, must be followed.
•
Ampul necks and vial stoppers are disinfected by wip-
ing with sufficient amounts of sterile 70% IPA to en-
sure that the critical sites remain wet for 10 seconds
and are allowed to dry.
•
Direct contact contamination of critical sites is mini-
mized by utilizing aseptic compounding.
•
Multiple-dose vials are labeled with the name of one
patient and a BUD and storage temperature range
based on manufacturer recommendations or published
literature. Single-dose extracts are not to be stored for
subsequent use after entry.
Nuclear pharmacies are regulated by the Nuclear Regu-
latory Commission as well as other applicable pharmacy laws
and regulations. USP chapter 823, Radiopharmaceuticals for
Positron Emission Tomography,
79
provides the standards for
production facilities. Once the components for use in posi-
tron emission tomography are released as finished prepa-
rations, handling, manipulation, and use are considered
compounding by USP chapter 797
15
and for the purposes
of these guidelines. Low-risk-level radiopharmaceuticals are
those compounded from sterile components with a volume
of less than 100 mL for a single-dose injection or no more
than 30 mL taken from a multiple-dose container.
15
These
radiopharmaceutical preparations must be compounded in
an ISO Class 5 PEC within an ISO Class 8 environment.
Non-radiopharmaceuticals compounded in nuclear
pharmacies must be compounded under full USP 797 compli-
ant conditions. The radiopharmaceutical exemption in USP
chapter 797
15
does not apply to non-radiopharmaceuticals.
The concept of limiting radiation exposure to a level
that is as low as reasonably achievable (ALARA) must be
adhered to for handling, compounding, and visual inspec-
tion of products. Technetium-99m/molybdenum-99 gen-
erator system operations and storage must occur within an
ISO Class 8 environment and must further comply with
all manufacturer’s recommendations and federal and state
regulations. Manufacturer’s guidelines for BUDs should be
followed for radiopharmaceutical multiple-dose vials that

126 Drug Distribution and Control: Preparation and Handling–Guidelines
are compounded with technetium-99m and exposed to ISO
Class 5 conditions with no direct contact contamination.
Personnel Compounding Competency
Touch contamination remains the primary cause of micro-
bial contamination in sterile compounding.
71,80
For this rea-
son, personnel training and assessment of competency are
of utmost importance to ensure the lowest possible risk for
contamination due to human error. For low- and medium-
risk operations, training and competency assessment are re-
quired initially upon hire or upon transfer to compounding
responsibilities, and again at least every 12 months for all
staff involved in the compounding of sterile products. High-
risk operations require more frequent assessments, and staff
must be evaluated upon hire or transfer and again at least
every six months.
As part of the training competency assessment, a writ-
ten test that evaluates knowledge about proper compound-
ing SOPs, aseptic technique, cleaning and garbing, environ-
mental monitoring, calculations, risk levels and BUDs, and
quality assurance principles must be successfully completed.
Thresholds for passing the written examination should be set
by the facility. While written tests assess knowledge, hands-
on observation of daily duties assesses for proper technique.
Personnel should be able to demonstrate at least the follow-
ing in a hands-on, witnessed assessment, as applicable to
their compounding responsibilities:
•
Proper hand hygiene technique (see Appendix III of
USP chapter 797
15
for a sample assessment form).
•
Proper gloving and garbing technique, including suc-
cessful glove fingertip test (see Appendix III of USP
chapter 797
15
for a sample assessment form).
•
Proper aseptic technique, including successful media-
fill test (see Appendix IV of USP chapter 797
15
for a
sample assessment form).
•
Proper cleaning and disinfecting procedures, including
successful surface sampling test (see Appendix V of
USP chapter 797
15
for a sample assessment form and
Appendix II for information about cleaning products).
•
Competency in the compounding of hazardous drugs.
•
Competency in the compounding of allergen extracts.
•
Competency in the compounding of radiopharmaceu-
ticals.
•
Competency in the use of sterile devices, such as filter
needles, injection port adapters, sterile fluid transfer
devices, and CSTDs.
•
Competency in the use of pharmacy compounding de-
vices.
•
Ability to fill pump reservoirs.
•
Competency to perform end-product testing and steril-
ization.
USP chapter 797
15
requires specific assessments to be
completed using sterile nutrient agar growth media to test
for potential contamination. Personnel-specific examples of
this type of testing include media-fill testing of aseptic tech-
nique and glove fingertip testing of compounding personnel.
Media-Fill Testing
As described in USP chapter 797, the media-fill component
of personnel assessment provides an objective evaluation of
aseptic technique.
15
Media-fill tests should be customized to
mimic the most challenging preparations compounded by
personnel on a regular basis in a specific facility. Testing
should occur at least every 12 months for personnel who
compound low- and medium-risk preparations, while testing
at least every 6 months is required for personnel involved
in compounding high-risk preparations. The actual test-
ing should take place under conditions that reflect realistic
workflow, such as the end of a shift, to simulate a worst-
case scenario environment for compounding sterile prepa-
rations. Once started, the test should be completed without
interruption. Fluid culture media are available commercially
for low- and medium-risk evaluations. High-risk assess-
ments may utilize nonsterile nutrient medium in a powder
form, which may be diluted and sterilized by filter methods.
Finished tests should be incubated per manufacturer’s rec-
ommendations. If incubators are in the pharmacy, they must
be placed outside the sterile compounding area. Ideally, the
facility’s microbiology services should incubate and read
the tests, providing an independent evaluation by qualified
individuals. Turbidity in the culture media signifies failure
of the media-fill testing and requires retesting of compound-
ing personnel. Personnel who fail these tests will require
re-training and may not compound sterile preparations until
tests have been repeated with successful results.
Glove Fingertip Testing
Three sets of glove fingertip evaluations must be completed
with no growth prior to personnel being allowed to com-
pound sterile preparations. This initial testing involves com-
pounding personnel completing all necessary hand hygiene
and garbing procedures (with the exception of applying
sterile 70% IPA to gloves). Immediately upon completion
of these procedures, the glove fingertip and thumb samples
from each hand are placed on sterile nutrient agar plates. The
samples should be incubated (such as by the facility’s mi-
crobiology personnel) according to manufacturer standards.
This test must be successfully completed three times ini-
tially, then at least every 12 months. Personnel compound-
ing high-risk products must successfully complete the test
at least every 6 months. Suggested thresholds for contami-
nation limits can be found in Table 6. Patterns of failures
(personnel, media, or facility) must be evaluated as part of
the facility’s quality assurance plan. Qualified microbiology
personnel and the facility’s infection control practitioner
should be consulted.
Growth Media Requirements
Sterile nutrient agar for media-fill testing, plates for fin-
gertip testing, and surface testing materials are available
from multiple vendors. USP chapters 797 and 1116 provide
specifications and requirements.
15,81
The media-fill testing
growth media and viable airborne particle plates should uti-
lize a Soybean-Casein Digest medium, which may also be
sold as Tryptic Soy Agar/Broth. The agar plates for glove
fingertip testing and surface testing should utilize general
nutrient agar with neutralizing agents such as lecithin and
polysorbate 80.
SOP Development
SOPs are documents containing detailed, step-by-step in-
structions on how to perform a task or procedure so that all

Drug Distribution and Control: Preparation and Handling–Guidelines 127
personnel consistently perform the task or procedure in the
same manner. SOPs are part of a good quality assurance pro-
gram within the pharmacy. They provide assurance that:
•
Equipment and facilities are properly maintained in
good working order.
•
Personnel are properly educated, trained, and evaluated.
•
Supplies are received, stored, and disposed of properly
and meet compendial standards.
•
All tasks and procedures are performed uniformly and
documented.
There are several components that should be included
in an SOP:
•
Title—should clearly identify the task.
•
SOP number—an internal department number as-
signed by the organization to identify it.
•
Author(s)—the name of the person or persons who
write the SOP so that problems and revisions can be
addressed.
•
Date effective—date when the SOP is implemented
into the compounding routine.
•
Authorization signature—person or committee that
approves the SOP.
•
Responsibility—person- or persons-in-charge who are
responsible for making sure that the SOP is performed
properly.
•
Purpose of the procedure—brief explanation of why
the SOP is necessary or being implemented.
•
Equipment and supplies required—list of equipment
and supplies needed to perform the SOP.
•
Procedure—detailed step-by-step explanation that can
be easily followed by different individuals with the
same results. The instructions should be concise to
minimize any required interpretation.
•
References—references should be listed to support the
implementation and use of the SOP.
•
Documentation form—easily accessible written re-
cord or log that demonstrates that the SOP is being
performed routinely and properly.
•
Revision—documentation of the date that an SOP has
been reviewed and the name of the reviewer.
USP chapter 797
15
lists and recommends SOPs and
should be reviewed to guide the pharmacy department in de-
veloping, writing, and implementing SOPs. There should be
SOPs written to address tasks or procedures in the following
general categories:
•
Personnel—training, education, skills, competency
evaluations, and responsibilities.
•
Facilities—access, cleaning, maintenance, use, moni-
toring, and testing.
•
Equipment—calibration, maintenance, cleaning, certi-
fication, verification, and use.
•
Supplies—ordering, storing, certification, inspection,
and disposal.
•
Compounding procedures—preparation of various
sterile compounded medications (e.g., batches, total
parenteral nutrition, hazardous drugs, epidural, pa-
tient-controlled analgesia, or ophthalmics), formulas,
assigning BUDs, handling, and packaging.
•
Safety—injuries, hazardous spills, and accidental ex-
posures.
•
Quality assurance—inspection of CSPs, testing of
CSPs, BUDs, delivery, and storage of final CSPs, pa-
tient monitoring, adverse event reporting, and person-
nel and environmental monitoring.
•
Administration—record keeping and management.
All significant procedures performed in a pharmacy
should be covered by SOPs and documentation. These pro-
cedures should be routinely reviewed and modified for im-
provements at least annually.
Quality Assurance Program
The purpose of a quality assurance program is to provide
a mechanism for monitoring, evaluating, correcting, and
improving activities and processes. A quality assurance pro-
gram should review and analyze objective data and use these
data to develop action plans. Facilities should actively work
to correct problems detected and improve activities and pro-
cesses as needed. Any plan designed to correct problems
should include follow-up parameters to make certain actions
were taken and were effective.
Activities and processes that are identified based on
their high frequency, high risk, or problem-prone nature
should have specific monitoring and evaluation criteria as-
signed for objective and measurable assessment. The quality
assurance program should encompass any and all activities
that are included in previous sections of this document as
elements which should be assessed and documented. This
includes, but is not limited to:
•
Personnel training and assessment
•
Environmental monitoring
•
Equipment calibration and maintenance
Specific quality assurance measures, pursuant to each
risk level compounded in a facility, include routine cleaning
and disinfection and air quality testing, visual confirmation
of proper garbing procedures, review of all orders and prep-
arations to ensure accuracy of compounded products, and
visual inspection of final CSPs to confirm the absence of
particulate matter or leakage.
A critical part of any quality assurance program is
proper documentation, corrective action, and follow-up.
Institutions must determine how results will be reported
and evaluated, including development of action limits and
thresholds. Thresholds and follow-up mechanisms must
be in place prior to initiating a quality assurance program
or immediately after collecting initial benchmark data.
Responsible persons for completing these tasks should be
identified and trained, if necessary, in the proper execution
of the quality assurance plan. Results of monitoring and
measurements should be reported within and outside of the
department responsible for compounding practices to com-
mittees such as Infection Control and Quality Improvement.
If corrective action is needed, the problem should be
resolved as soon as possible. Assessment of problems with
compounding errors, evident contamination during prepara-
tion, quarantine, or patterns of personnel or environmental
monitoring outside the established parameters require for-

128 Drug Distribution and Control: Preparation and Handling–Guidelines
mal follow-up. A root cause analysis, including participation
by other facility experts such as infection control personnel,
should be completed.
82
For situations needing more time for
corrective measures, an action plan should be developed and
followed. Indicators and effectiveness of the quality assur-
ance program should be reassessed annually.
New technologies, procedures, and policies should be
incorporated on an as-needed basis. A failure mode and ef-
fects analysis of new techniques can serve as a valuable pro-
active assessment of the ease and value prior to introduction
into the compounding process.
83
References
1. Hasegawa GR. Caring about stability and compatibil-
ity. Am J Hosp Pharm. 1994; 51:1533–4. [editorial]
2. Stability considerations in dispensing practice (general
information chapter 1191). In: The United States phar-
macopeia, 34th rev., and The national formulary, 29th
ed. Rockville, MD: The United States Pharmacopeial
Convention; 2011:742–5.
3. Guynn JB Jr, Poretz DM, Duma RJ. Growth of various
bacteria in a variety of intravenous fluids. Am J Hosp
Pharm. 1973; 30:321–5.
4. Selenic D, Dodson DR, Jensen B, et al. Enterobacter
cloacae bloodstream infection in pediatric patients
traced to a hospital pharmacy. Am J Health-Syst
Pharm. 2003; 60:1440–6.
5. Hughes CF, Grant AF, Lick BD, et al. Cardioplegic
solution: a contamination crisis. J Thorac Cardiovasc
Surg. 1986; 91:296–302.
6. Dugleaux G, Coutour XL, Hecquard C, et al.
Septicemia caused by contaminated parenteral nutri-
tion pouches: the refrigerator as an unusual cause. J
Parenter Enteral Nutr. 1991; 15:474–5.
7. Sachs GS. Microbial contamination of parenteral
nutrition—How could it happen? J Parenter Enteral
Nutr. 2011; 35:432.
8. Traynor K. Meningitis outbreak challenges hos-
pital pharmacies. Am J Health-Syst Pharm. 2012;
69:2024–6.
9. Pierce LR, Gaines A, Varricchio R, et al. Hemolysis
and renal failure associated with use of sterile water
for injection to dilute 25% human albumin solution.
Am J Health-Syst Pharm. 1998; 55:1057,1062,1070.
10. Flynn EA, Pearson RE, Barker KN. Observational
study of accuracy in compounding i.v. admixtures
at five hospitals. Am J Health-Syst Pharm. 1997;
54:904–12.
11. Morris AM, Schneider PJ, Pedersen CA, et al. National
survey of quality assurance activities for pharmacy-
compounded sterile preparations. Am J Health-Syst
Pharm. 2003; 60:2567–76.
12. Santell JP, Kamalich RF. National survey of quality as-
surance activities for pharmacy-prepared sterile prod-
ucts in hospitals and home infusion facilities—1995.
Am J Health-Syst Pharm. 1996; 53:2591–605.
13. Kastango ES, Douglass K. Improving the manage-
ment, operations and cost effectiveness of sterile-prod-
uct compounding. Int J Pharm Compd. 1999; 3:253–8.
14. American Society of Health-System Pharmacists.
ASHP Guidelines on Quality Assurance for Pharmacy-
Prepared Sterile Products. Am J Health-Syst Pharm.
2000; 57:1150–69.
15. Pharmaceutical compounding—sterile preparations
(general information chapter 797). In: The United
States Pharmacopeia, 36th rev., and the National
Formulary, 31 ed. Rockville, MD: The United States
Pharmacopeial Convention; 2013: 361–98.
16. American Society of Health-System Pharmacists.
ASHP guidelines on outsourcing sterile compounding
services. Am J Health-Syst Pharm. 2010; 67:757–65.
17. ASHP Discussion Guide on USP Chapter <797>; 2008.
www.ashp.org/s_ashp/docs/files/DiscGuide797-2008.
pdf (accessed 2012 Dec 28).
18. Food and Drug Administration Modernization
Act of 1997, Pub. L. No. 105-115, 111 Stat. 2296.
www.fda.gov/RegulatoryInformation/Legislation/
FederalFoodDrugandCosmeticActFDCAct/
SignificantAmendmentstotheFDCAct/FDAMA/de-
fault.htm (accessed 2012 Dec 28).
19.
Office of Regulatory Policy and Office of Compliance,
Center for Drug Evaluation and Research (CDER),
Food and Drug Administration. Guidance for FDA staff
and industry. Compliance policy guides manual. Sec.
460.200. Pharmacy compounding. www.fda.gov/ICECI/
ComplianceManuals/CompliancePolicyGuidanceManual/
ucm074398.htm (accessed 2012 Dec 28).
20. Sterile drug products for home use (general informa-
tion chapter 1206). In: The United States pharma-
copeia, 23rd rev., and The national formulary, 19th
ed. Rockville, MD: The United State Pharmacopeial
Convention; 1995:2130–43.
21. Douglas K, Kastango E, Cantor P. State regulations
impact USP <797> compliance. Pharm Purch Prod.
2012; 9:S18–S21.
22. National Association of Boards of Pharmacy. Model
State Pharmacy Act and Model Rules of the National
Association of Boards of Pharmacy. www.nabp.net/
publications/model-act/ (accessed 2012 Dec 28).
23. CMS Hospital Conditions of Participation and Interpretative
Guidelines, Centers for Medicare and Medicaid Services
(CMS), Department of Health and Human Services.
State Operations Manual Appendix A—Survey Protocol,
Regulations and Interpretive Guidelines for Hospitals,
Rev 78 (12-22-11). Washington, DC: Government Printing
Office; 2011. www.cms.gov/Regulations-and-Guidance/
Guidance/Manuals/downloads/som107ap_a_hospitals.pdf
(accessed 2012 Dec 28).
24. Comprehensive accreditation manual for hospitals: the
official handbook. Oakbrook Terrace, IL: The Joint
Commission; 2012.
25. American Osteopathic Association. Healthcare
Facilities Accreditation Program, 2009 Accreditation
Requirements for Healthcare Facilities; 2009.
26. DNV Healthcare. National Integrated Accreditation
for Healthcare Organizations (NIAHO), Interpretive
Guidelines and Surveyor Guidance, Revision 10;
2012.
27. National Institute for Occupational Safety and Health
[NIOSH]. NIOSH List of Antineoplastic and Other
Hazardous Drugs in Healthcare Settings 2012. www.
cdc.gov/niosh/docs/2012-150/ (accessed 2012 Dec
28).

Drug Distribution and Control: Preparation and Handling–Guidelines 129
28. American Society of Health-System Pharmacists.
ASHP guidelines on handling hazardous drugs. Am J
Health-Syst Pharm. 2006; 63:1172–93.
29. National Institute for Occupational Safety and Health
[NIOSH]. NIOSH alert: preventing occupational ex-
posure to antineoplastic and other hazardous drugs
in health care settings; 2004. www.cdc.gov/niosh/
docs/2004-165/#g (accessed 2012 Dec 28).
30. National Institute for Occupational Safety and Health
[NIOSH]. Personal protective equipment for health
care workers who work with hazardous drugs (NIOSH
document 2009-16). www.cdc.gov/niosh/docs/wp-
solutions/2009-106/ (accessed 2012 Dec 20).
31. American Society of Hospital Pharmacists. ASHP tech-
nical assistance bulletin on pharmacy-prepared oph-
thalmic products. Am J Hosp Pharm. 1993; 50:1462–3.
32. American Society of Health-System Pharmacists.
ASHP guidelines on the safe use of automated com-
pounding devices for the preparation of parenteral
nutrition admixtures. Am J Health-Syst Pharm. 2000;
57:1343–8.
33. American Society of Health-System Pharmacists.
ASHP guidelines: minimum standard for pharmacies in
hospitals. Am J Health-Syst Pharm. 2013; 70:1619–30.
34. ASPEN Task Force for the Revision of Safe Practices
for Parenteral Nutrition: Mirtallo J, Canada T, Johnson
D, et al. Safe Practices for Parenteral Nutrition. J
Parenter Enteral Nutr. 2004; 28(6):S39–S70.
35. Bankhead R, Boullata J, Brantley S, et al. Enteral nu-
trition practice recommendations. J Parenter Enteral
Nutr. 2009; 33:122–67.
36. Institute for Safe Medication Practices [ISMP]. 2004
ISMP medication safety self assessment for hospitals.
Huntington Valley, PA: ISMP; 2004. www.ismp.org/
selfassessments/Hospital/2004Hospsm.pdf (accessed
2012 Dec 28).
37. Institute for Safe Medication Practices [ISMP].
Proceedings from the ISMP Sterile Preparation
Compounding Safety Summit: Guidelines for SAFE
Preparation of Sterile Compounds; 2012. www.ismp.
org/tools/guidelines/IVSummit/IVCGuidelines.pdf
(accessed 2012 Dec 28).
38. Infusion Nurses Society. Infusion nursing standards of
practice. J Infus Nurs. 2011; 34(1S):S1–S110.
39. Controlled Environment Testing Association [CETA].
CETA certification guide for sterile compounding fa-
cilities (CAG-003-2006) (revised January 31, 2012).
www.cetainternational.org/reference/CAG-003-
2006v11.pdf (accessed 2012 Dec 28).
40. Controlled Environment Testing Association [CETA].
Servicing hazardous drug compounding primary engi-
neering controls (CAG-005-2007). www.cetainterna-
tional.org/reference/CAG005-v15.pdf (accessed 2012
Dec 28).
41. Controlled Environment Testing Association [CETA].
Application guide for the use of surface decontami-
nants in biosafety cabinets (CAG-004-2007). www.
cetainternational.org/reference/CAG0042007i.pdf
(accessed 2012 Dec 28).
42. Controlled Environment Testing Association [CETA].
Compounding isolator testing guide (CAG-002-2006) (re-
vised December 2008). www.cetainternational.org/ref-
erence/CETACompoundingIsolatorTestingGuide2006.
pdf (accessed 2012 Dec 28).
43. Controlled Environment Testing Association [CETA].
Application guide for the use of compounding isola-
tors in compounding sterile preparations in healthcare
facilities (CAG-001-2005) (revised December 2008).
www.cetainternational.org/reference/Applications
GuideBarrierIsolator-CAG-001-2005.pdf (accessed
2012 Dec 28).
44. Controlled Environment Testing Association [CETA].
CETA Certification Matrix for Sterile Compounding
Facilities (CAG-008-2010) (updated January 2012).
www.cetainternational.org/reference/CAG-008-
2010v2.pdf (accessed 2012 Dec 20).
45. Controlled Environment Testing Association
[CETA]. CETA Certification Application Guide USP
<797> Viable Environmental Sampling & Gowning
Evaluation (CAG-009-2011v3). www.cetainterna-
tional.org/reference/CAG-009v3.pdf (accessed 2012
Dec 28).
46. Centers for Disease Control and Prevention [CDC].
Guidelines for hand hygiene in healthcare settings.
MMWR 2002; 51(RR16):1–45. www.cdc.gov/mmwr/pre-
view/mmwrhtml/rr5116a1.htm (accessed 2012 Dec 28).
47. O’Grady NP, Alexander M, Burns LA, et al. 2011
Guidelines for the Prevention of Intravascular Catheter-
Related Infections. Centers for Disease Control and
Prevention, 2012. www.cdc.gov/hicpac/pdf/guide
-
lines/bsi-guidelines-2011.pdf (accessed 2012 Dec 28).
48. Centers for Disease Control and Prevention [CDC].
Guidelines for Environmental Infection Control in
Healthcare Facilities. MMWR. 2003; 52(RR10):1–42.
www.cdc.gov/mmwr/preview/mmwrhtml/rr5210a1.
htm (accessed 2012 Dec 28).
49. Centers for Disease Control and Prevention, National
Center for Emerging and Zoonotic Infectious Diseases
(NCEZID), Division of Healthcare Quality Promotion
(DHQP). Protect Patients Against Preventable Harm from
Improper Use of Single-Dose/Single-Use Vials. www.
cdc.gov/injectionsafety/CDCposition-SingleUseVial.
html (accessed 2012 Dec 28).
50. Centers for Medicare & Medicaid Services [CMS].
Single use of single dose/single use medications to
prevent healthcare-associated infections. www.cms.
gov/Medicare/Provider-Enrollment-and-Certification/
SurveyCertificationGenInfo/Policy-and-Memos-to-
States-and-Regions-Items/Survey-and-Cert-Letter-12-35.
html (accessed 2012 Dec 28).
51. Dolan SA, Felizardo G, Barnes S, et al. APIC position
paper: Safe injection, infusion, and medication vial
practices in health care. Am J Infect Control. 2010;
38:167–72.
52. Chapter 17. Recommended Practices for Medication
Safety. In: Conner R, Blanchard J, Burlingame B, et
al. (eds). Perioperative Standards and Recommended
Practices. Denver, CO: Association of periOperative
Registered Nurses (AORN); 2012:251–300.
53. Buchanan EC, Schneider PJ. Compounding sterile
preparations, 3rd ed. Bethesda, MD: American Society
of Health-System Pharmacists; 2009.
54. American Society of Health-System Pharmacists
[ASHP]. Basics of aseptic compounding technique.
Bethesda, MD: ASHP; 2006.

130 Drug Distribution and Control: Preparation and Handling–Guidelines
55. Davis K, Sparks J. Getting started in aseptic com-
pounding. Bethesda, MD: American Society of
Health-System Pharmacists; 2008.
56. Kienle PC. Compounding sterile preparations:
ASHP video guide to chapter <797>. Bethesda, MD:
American Society of Health-System Pharmacists;
2009.
57. U.S. Department of Health and Human Services, Food
and Drug Administration, Office of Regulatory Affairs.
Guidance for industry: sterile drug products produced
by aseptic processing—current good manufacturing
practice. September 2004. www.fda.gov/downloads/
Drugs/GuidanceComplianceRegulatoryInformation/
Guidances/UCM070342.pdf (accessed 2012 Dec 28).
58. General notices and requirements. In: The United
States pharmacopeia, 34th rev., and The national for-
mulary, 29th ed. Rockville, MD: The United States
Pharmacopeial Convention; 2011:11–12.
59. Pharmaceutical compounding—nonsterile prepa-
rations (general information chapter 795). In: The
United States pharmacopeia, 34th rev., and The na-
tional formulary, 29th ed. Rockville, MD: The United
States Pharmacopeial Convention; 2011:330–6.
60. Trissel LA. Handbook on injectable drugs. 17th ed.
Bethesda, MD: American Society of Health-System
Pharmacists; 2013.
61. Bing CD, Nowobilski-Vasilios A. Extended stability
for parenteral drugs. 5th ed. Bethesda, MD: American
Society of Health-System Pharmacists; 2013:321.
62. Personal communication from Hospira. December 16,
2008.
63. Personal communication from Baxter. December 18,
2008.
64. Personal communication from B. Braun. December
26, 2008.
65. Injections (general information chapter 1). In: The
United States pharmacopeia, 34th rev., and The na-
tional formulary, 29th ed. Rockville, MD: The United
States Pharmacopeial Convention; 2011:33–8.
66. Centers for Disease Control and Prevention, National
Center for Emerging and Zoonotic Infectious Diseases
(NCEZID), Division of Healthcare Quality Promotion
(DHQP). Safe Injection Practices to Prevent Transmission
of Infections to Patients. www.cdc.gov/injectionsafety/
IP07_standardPrecaution.html (accessed 2013 Jan 30).
67. Sterility tests (general information chapter 71). In: The
United States pharmacopeia, 34th rev., and The na-
tional formulary, 29th ed. Rockville, MD: The United
States Pharmacopeial Convention; 2011:65–70.
68. Bacterial endotoxins test (general information chapter
85). In: The United States pharmacopeia, 34th rev.,
and The national formulary, 29th ed. Rockville, MD:
The United States Pharmacopeial Convention; 2011:
78–82.
69. Pyrogen test (general information chapter 151). In: The
United States pharmacopeia, 34th rev., and The na-
tional formulary, 29th ed. Rockville, MD: The United
States Pharmacopeial Convention; 201
1:115–7.
70. Oversight of Care, Treatment, and Services
Provided through Contractual Agreement (Standard
LD.04.03.09). Comprehensive accreditation manual
for hospitals: the official handbook. Oakbrook Terrace,
IL: The Joint Commission; 2012.
71. Trissel LA, Gentempo JA, Saenz LM, et al. Effect of
two work practice changes on the microbial contami-
nation rates of pharmacy-compounded sterile prepara-
tions. Am J Health-Syst Pharm. 2007; 64:837–41.
72. Sessink PJ, Boer KA, Scheefhals AP, et al.
Occupational exposure to antineoplastic agents at sev-
eral departments in a hospital: environmental contami-
nation and excretion of cyclophosphamide and ifos-
famide in urine of exposed workers. Int Arch Occup
Environ Health. 1992; 64:105–12.
73. Kiffmeyer TK, Ing KG, Schoppe G. External con-
tamination of cytotoxic drug packing: safe handling
and cleaning procedures. J Oncol Pharm Pract. 2000;
6:13.
74. Connor TH, Sessink PJ, Harrison BR et al. Surface
contamination of chemotherapy drug vials and evalu-
ation of new vial-cleaning techniques: results of three
studies. Am J Health-Syst Pharm. 2005; 62:475–84.
75. Kastango ES, Wagner JT, Kastango KB, et al.
Generation of particulate matter during handling
of needle and syringe packaging. Am J Health-Syst
Pharm. 2008; 65:1443–50.
76. OSHA Hazard Communication Standard (revised
2012). www.osha.gov/dsg/hazcom/index.html. (ac-
cessed 2012 Dec 28).
77. National Institute for Occupational Safety and Health
[NIOSH]. Medical surveillance for healthcare workers
exposed to hazardous drugs. NIOSH Publication No.
2013-103; November 2012. www.cdc.gov/niosh/docs/
wp-solutions/2013-103/ (accessed 2012 Dec 28).
78. Joint Council of Allergy, Asthma, and Immunology.
Allergen Immunotherapy Extract Preparation
Guidelines. www.jcaai.org/file_
depot/0-10000000/
20000-30000/27387/folder/62846/
Allergen_Extract_
Preparation_Guidelines.pdf (accessed 2012 Dec 28).
79. Radiopharmaceuticals for positron emission tomog-
raphy—compounding (general information chap-
ter 823). In: The United States pharmacopeia, 34th
rev., and The national formulary, 29th ed. Rockville,
MD: The United States Pharmacopeial Convention,
2011:385–9.
80. Thomas M, Sanborn MD, Couldry R. I.V. admixture
contamination rates: Traditional practice site versus a
class 1000 cleanroom. Am J Health-Syst Pharm. 2005;
62:2386–92.
81. Microbiological evaluation of clean rooms and other
controlled environments (general information chapter
1116). In: The United States pharmacopeia, 34th rev.,
and The national formulary, 29th ed. Rockville, MD:
The United States Pharmacopeial Convention; 2011:
633–41.
82. Agency for Healthcare Research and Quality Patient
Safety Network (AHRQ PSNet). Patient safety primer:
root cause analysis. www.psnet.ahrq.gov/primer.
aspx?primerID=10 (accessed 2012 Dec 28).
83. DeRosier J, Stalhandske E, Bagian JP, et al. Using
health care failure mode and effects analysis: the VA
National Center for Patient Safety’s prospective risk
analysis system. Jt Comm J Qual Improv. 2002; 28:
248–67.
Approved by the ASHP Board of Directors on June 2, 2013.
Developed through the ASHP Council on Pharmacy Practice. These

Drug Distribution and Control: Preparation and Handling–Guidelines 131
guidelines supersede the ASHP Guidelines on Quality Assurance for
Pharmacy-Prepared Sterile Products dated April 27, 2000.
Patricia C. Kienle, M.P.A., FASHP, is gratefully acknowledged
for leading the development of and drafting substantial portions of
these guidelines. Linda F. McElhiney, Pharm.D.; Richard B. Osteen,
D.Ph.; Ed Troell, M.B.A.; Fred Massoomi, Pharm.D., FASHP;
Kathleen Sheehy, M.B.A.; and Angela T. Cassano, Pharm.D.,
BCPS, are also gratefully acknowledged for their contributions to
these guidelines.
ASHP gratefully acknowledges the following individuals for review-
ing these guidelines (review does not imply endorsement): Caryn
D. Bing, M.S., FASHP; E. Clyde Buchanan, M.S., FASHP; Ryan
A. Forrey, Pharm.D., M.S.; Eric S. Kastango, M.B.A., FASHP; Lee
B. Murdaugh, Ph.D.; Daryl McCollum, Pharm.D.; Luci A. Power,
M.S.; Philip J. Schneider, M.S., FASHP; and James T. Wagner.
Copyright © 2014, American Society of Health-System Pharmacists,
Inc. All rights reserved.
The bibliographic citation for this document is as follows: American
Society of Health-System Pharmacists. ASHP Guidelines on
Compounding Sterile Preparations. Am J Health-Syst Pharm. 2014;
71:145–66.
